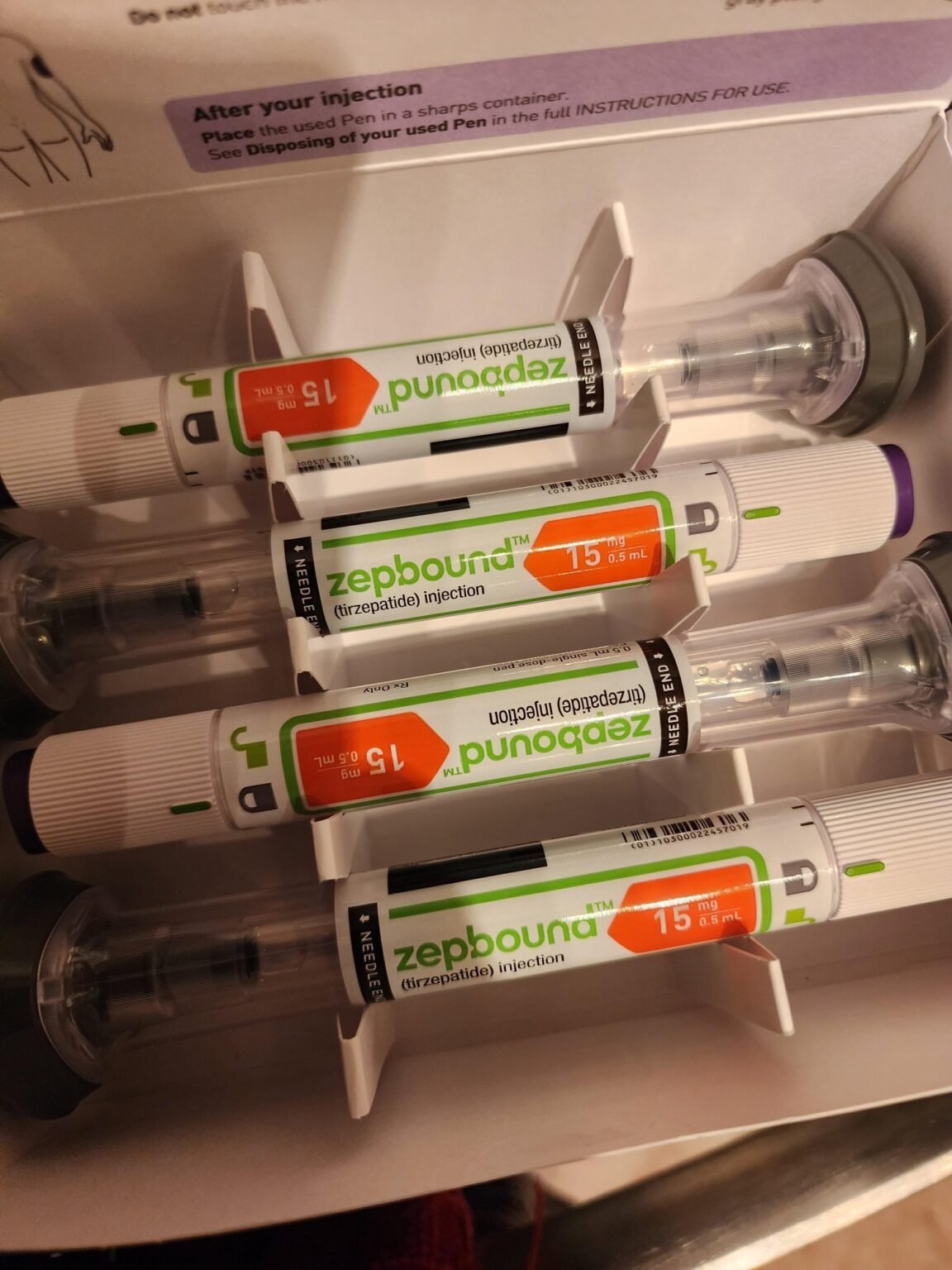
Zepbound
£130.00 – £455.00Price range: £130.00 through £455.00
Zepbound
Zepbound (tirzepatide) is a once-weekly injectable prescription medicine for chronic weight management. It’s a dual GIP/GLP-1 receptor agonist that helps reduce appetite and food intake. Used with diet and exercise, it can lead to significant weight loss for adults with obesity or weight-related health conditions.
SKU: N/A
Category: Weight Loss
Zepbound
Zepbound (Tirzepatide) for Chronic Weight Management
Zepbound (tirzepatide) is an injectable prescription medicine indicated as an adjunct to a reduced-calorie diet and increased physical activity for chronic weight management in adults. It is approved for use in individuals with an initial Body Mass Index (BMI) of:
* {30 kg/m} or greater (obesity), or
* {27 kg/m} or greater (overweight) in the presence of at least one weight-related comorbid condition (e.g., hypertension, dyslipidemia, type 2 diabetes mellitus, or obstructive sleep apnea).
Mechanism of Action: The Dual Incretin Agonist
Zepbound represents a breakthrough in weight management due to its novel dual mechanism of action. It is the first and only FDA-approved medication that acts as a dual agonist for both the glucose-dependent insulinotropic polypeptide (GIP) receptor and the glucagon-like peptide-1 (GLP-1) receptor. Both GIP and GLP-1 are naturally occurring incretin hormones that play a key role in metabolic regulation and appetite control.
By activating both receptors, tirzepatide works to promote weight loss and maintenance through several pathways:
Appetite and Satiety Regulation: The drug targets receptors in the brain to reduce appetite and decrease food intake, leading to reduced calorie consumption.
Delayed Gastric Emptying: It slows the movement of food from the stomach, which contributes to a feeling of fullness for a longer duration.
Metabolic Effects (in those with Type 2 Diabetes): While approved for weight management regardless of diabetes status, tirzepatide also stimulates glucose-dependent insulin secretion and lowers glucagon levels, leading to improved blood glucose control in patients with co-existing Type 2 Diabetes.
Dosage and Administration
Zepbound is administered as a once-weekly subcutaneous injection using a pre-filled, single-dose pen. The medication is dosed through an escalation schedule to minimize gastrointestinal adverse reactions.
Starting Dosage: {2.5 mg}$ injected subcutaneously once weekly for {4} weeks. This dose is for treatment initiation only and not for chronic weight management.
Titration: The dosage should be increased in {2.5 mg} increments after at least {4}$ weeks on the current dose.
Maintenance Dosages: The recommended maintenance dosages are {5 mg}, {10 mg}, or {15 mg} injected subcutaneously once weekly.
Maximum Dosage: The maximum recommended dosage is $\text{15 mg} once weekly.
Doses are available in {2.5 mg/0.5 mL}, {5 mg/0.5 mL}, {7.5 mg/0.5 mL}, {10 mg/0.5 mL}, {12.5 mg/0.5 mL}, and {15 mg/0.5 mL} pre-filled pens.
Clinical Efficacy: Demonstrated Weight Loss
In clinical trials (the SURMOUNT program), Zepbound demonstrated superior efficacy for chronic weight management compared to placebo and a single GLP-1 agonist.
In a 72-week trial evaluating adults with obesity or overweight without type 2 diabetes, participants treated with the highest dose of Zepbound {15 mg} achieved a mean weight reduction of up to $\text{20.9}\%$ from baseline.
In a head-to-head trial against a maximum-dose GLP-1 agonist {2.4 mg} semaglutide), Zepbound demonstrated superior weight loss, with participants achieving an average weight reduction of {20.2} with Zepbound vs. {13.7} with the comparator at {72} weeks.
Important Safety Information and Adverse Reactions
The most common adverse reactions reported in clinical trials, occurring primarily during dose escalation, were gastrointestinal-related, including nausea, diarrhea, vomiting, constipation, and dyspepsia. These events are generally mild to moderate and tend to decrease over time.
Boxed Warning: Zepbound carries a Boxed Warning regarding the risk of thyroid C-cell tumors. It is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Contraindications: Zepbound is also contraindicated in patients with a history of a serious hypersensitivity reaction to tirzepatide or any of its excipients.
Warnings and Precautions include risk of acute pancreatitis, acute gallbladder disease, acute kidney injury (secondary to gastrointestinal side effects), and hypoglycemia (especially when used with an insulin secretagogue or insulin). Due to its effect on gastric emptying, Zepbound has the potential to impact the absorption of concurrently administered oral medications.
| Pack Size | 1 Pen (1 Month Supply), 2 Pen (2 Months Supply), 3 Pen (3 Months Supply) |
|---|---|
| Strenght | 7.5mg, 10mg, 12.5mg, 15mg |
Be the first to review “Zepbound” Cancel reply
Related products
Weight Loss
£126.00 – £262.00Price range: £126.00 through £262.00
Weight Loss
£42.99
Weight Loss
£103.77 – £350.22Price range: £103.77 through £350.22
Weight Loss
£20.99 – £84.99Price range: £20.99 through £84.99
Weight Loss
£47.99 – £448.99Price range: £47.99 through £448.99
Weight Loss
£59.99 – £149.99Price range: £59.99 through £149.99
Weight Loss
£35.99
Weight Loss
£105.00 – £555.00Price range: £105.00 through £555.00







Reviews
There are no reviews yet.